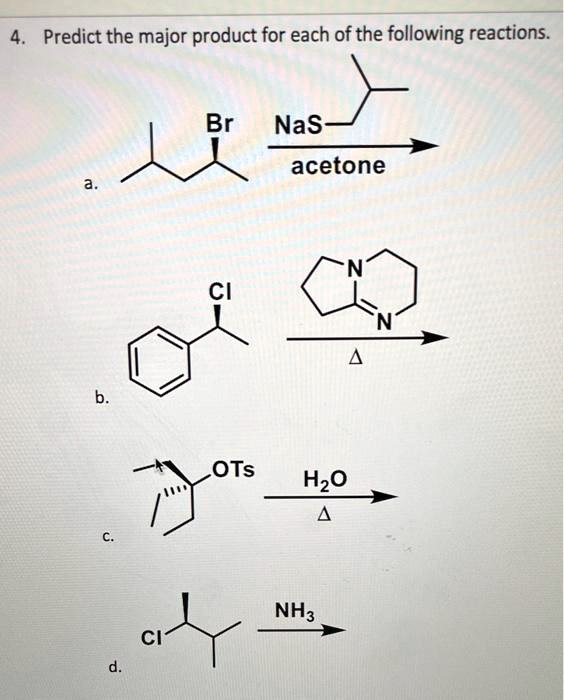
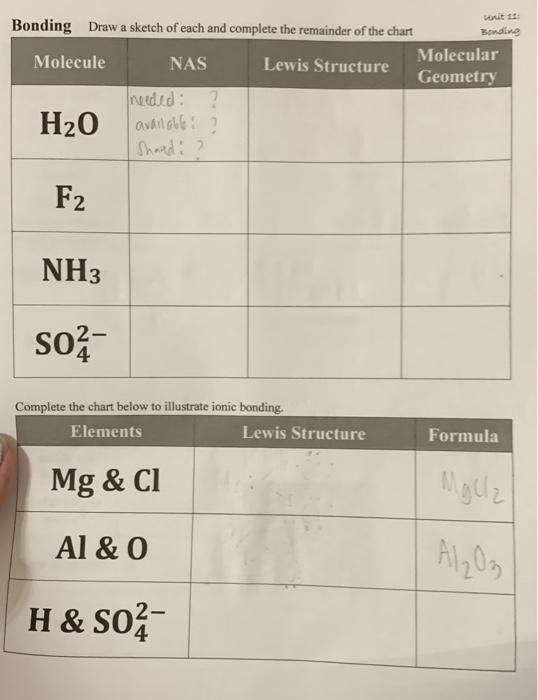
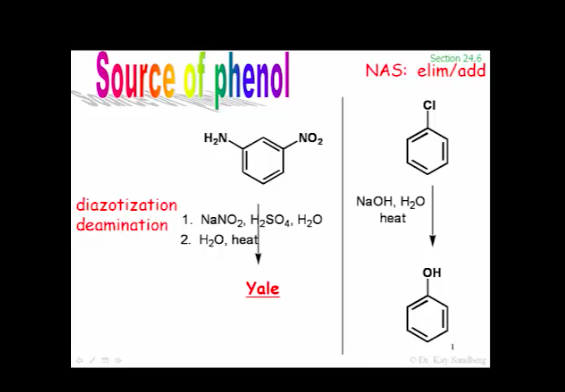
SOLVED: EXAMPLE 3 (MHR pg.646) Consider the reaction 2 Nas) + 2 H2O -> 2 NaOH(aq)+ H2(g) a) Calculate the standard cell potential for the galvanic cell b) Predict whether the reaction

Fórmula Química H2o Com Gotas Da água Nas Letras Ilustração do Vetor - Ilustração de fundo, respingo: 153532179