di-Sodium oxalate, 250 g, CAS No. 62-76-0 | Sodium Salts, Na | Salts for Analysis (p.a.) | Salts | Inorganic & Analytical Reagents | Chemicals | Carl Roth - France
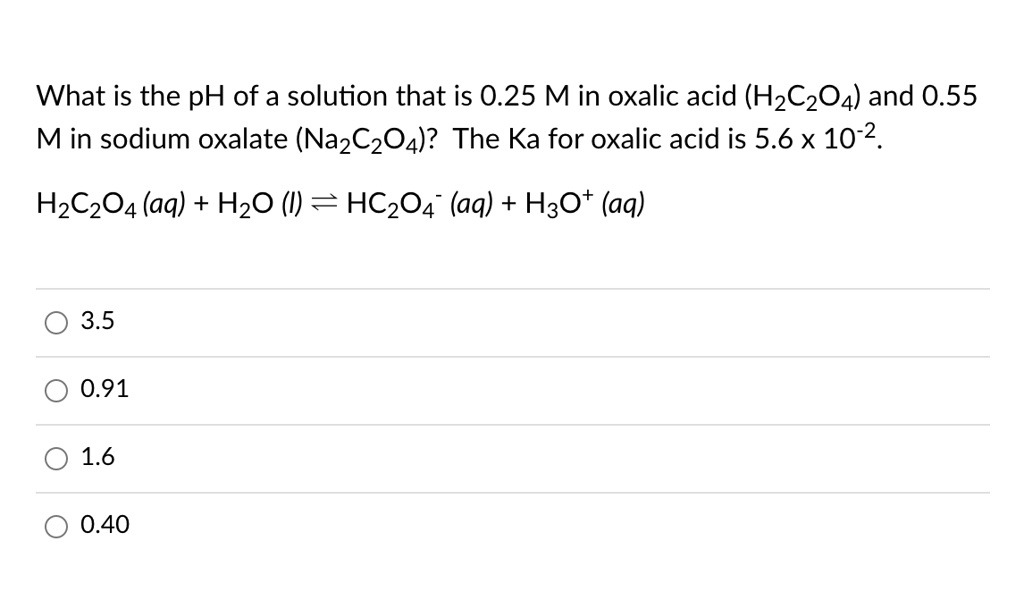
SOLVED: What is the pH of a solution that is 0.25 M in oxalic acid (H2C2O4) and 0.55 M in sodium oxalate (Na2C2O4)? The Ka for oxalic acid is 5.6 x 10^-2.

Solubility of Sodium Oxalate in Concentrated Electrolyte Solutions | Journal of Chemical & Engineering Data

Plot of solubility against reciprocal temperature for sodium oxalate.... | Download Scientific Diagram
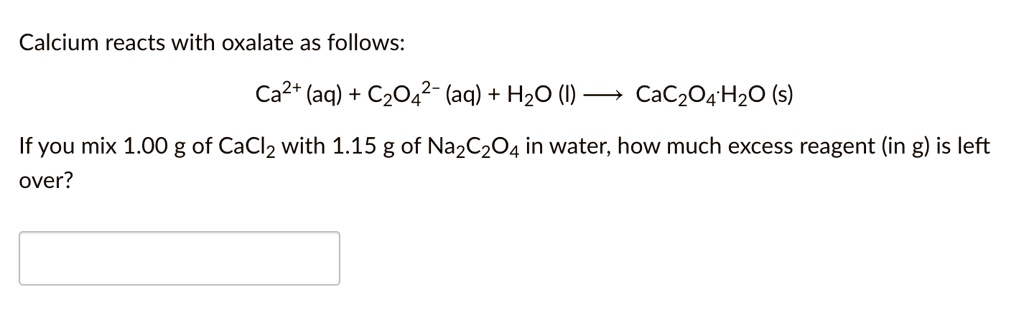
SOLVED: Calcium reacts with oxalate as follows: Ca2+ (aq) + C2O4^2- (aq) + H2O (l) CaC2O4·H2O (s) If you mix 1.00 g of CaCl2 with 1.15 g of Na2C2O4 in water, how

Mass spectrometric study of effluents during the temperature-programmed... | Download Scientific Diagram
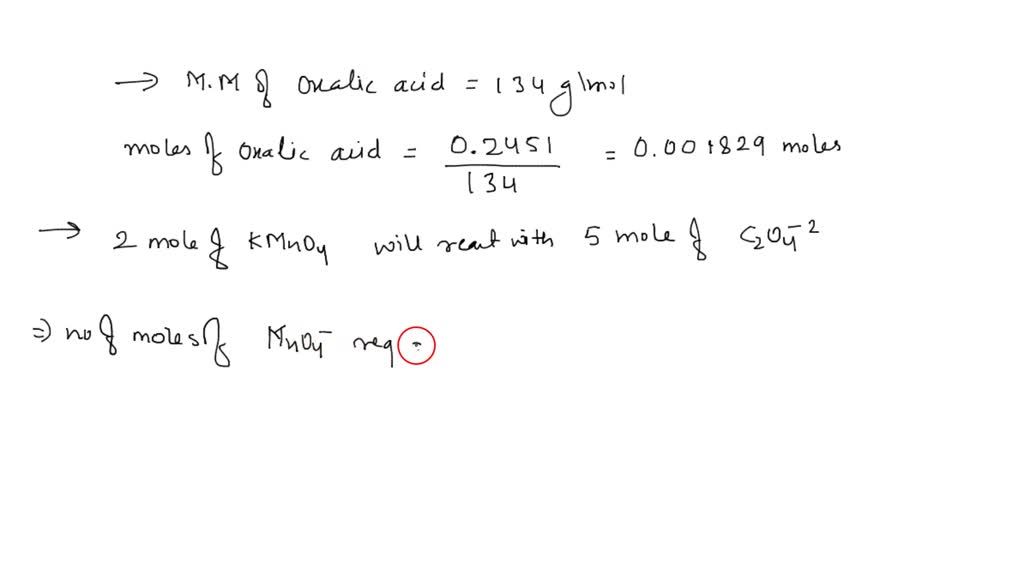
SOLVED: Potassium permanganate solutions may be standardized by titrating against the primary standard sodium oxalate following the reaction: 2 MnO4-(aq) + 5 C2O4^2-(aq) â†' 2 Mn^2+(aq) + 10 CO2(g) + 8 H2O(l).






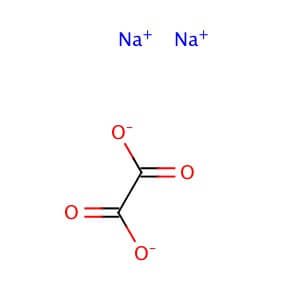






.jpg)


