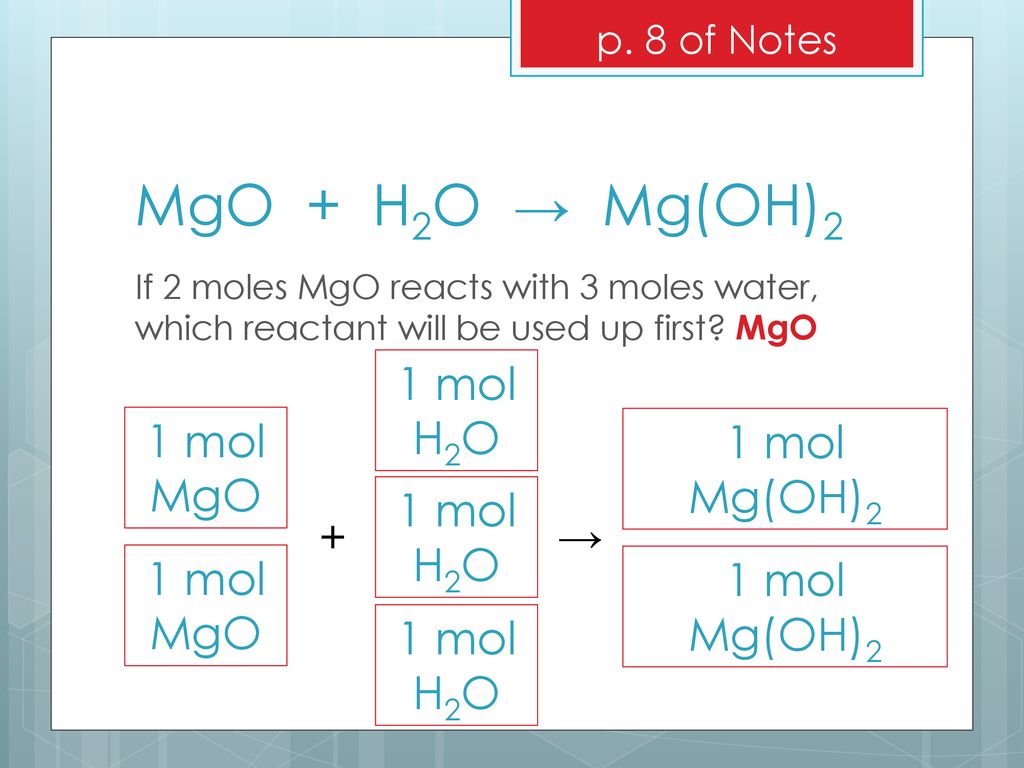
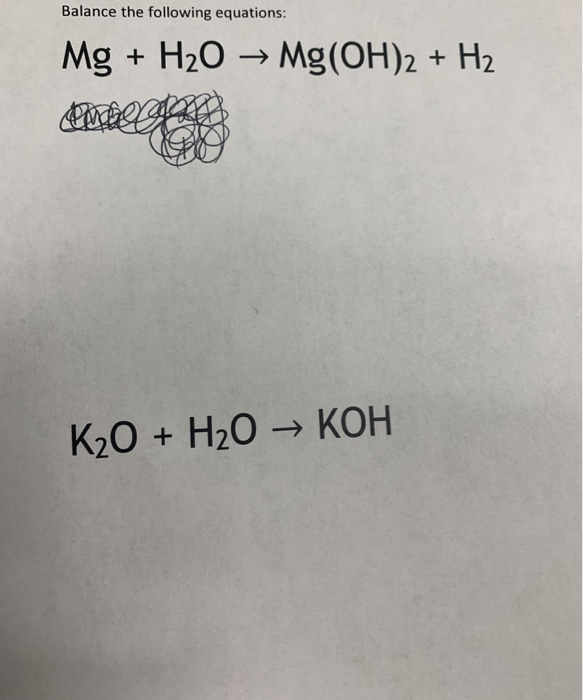SOLVED: Gaseous water (H2O) and solid magnesium oxide (MgO) are formed by the decomposition of solid magnesium hydroxide (Mg(OH)2). Write a balanced chemical equation for this reaction.

How to Balance Mg(OH)2 + HCl = MgCl2 + H2O (Magnesium Hydroxide + Hydrochlo | Balancing equations, Equations, Molecules

1) Mg3N2 + H2O ----------- + Mg(OH)2 + NH3 2) Pb(OH)2 + NaOH --------→ Na2PbO2 + H2O 3) C + H2SO4 ----------→ CO2 + H2O + SO2 4) Fe2O3 + CO ---------→ Fe + CO2 5) NH3 + Cl2 ----------→ HCl + NCl3 Dawritenfor matching the column

Solubility Equilibria in the System Mg(OH)2–MgCl2–H2O from 298 to 393 K | Journal of Chemical & Engineering Data

Solubility Equilibria in the System Mg(OH)2–MgCl2–H2O from 298 to 393 K | Journal of Chemical & Engineering Data