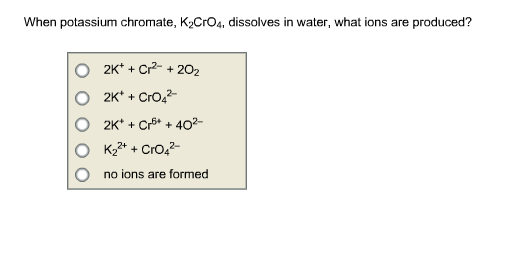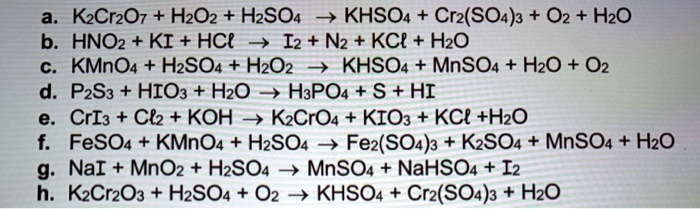
SOLVED: K2Cr2O7 + H2O2 + H2SO4 -> KHSO4 + Cr2(SO4)3 + O2 + H2O HNO2 + KI + HCl -> I2 + N2 + KCl + H2O KMnO4 + H2SO4 -> H2O2 +

A student titrates a 50.0 mL sample of chromic acid, H2CrO4, with 0.200 M KOH (aq). The titration reaches - Brainly.in
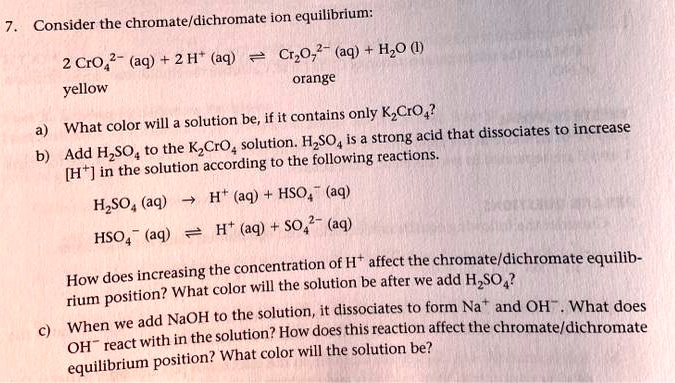
SOLVED: Consider the chromate/dichromate ion equilibrium: CrO4^2- (aq) + H2O (l) ⇌ 2 CrO4^2- (aq) + 2 H+ (aq). The solution will be orange-yellow if it contains only K2CrO4. What color will

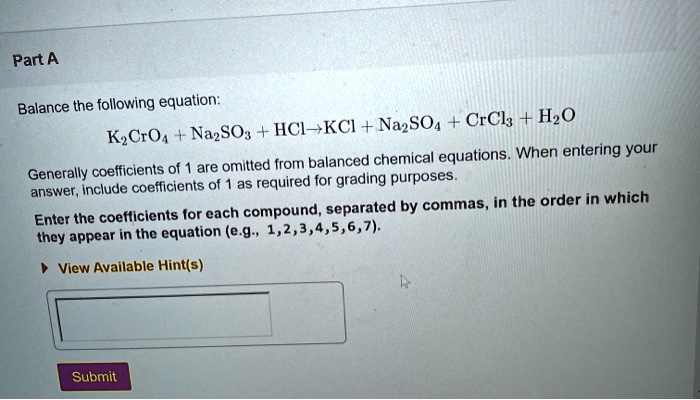
K2CrO4 + K2SO3 + H20 -> Cr(OH)3 + K2SO4+ KOH Uzupełnij współczynniki Bilansem jonowo-elektronowym. - Brainly.pl

SOLVED: Potassium chromate (K2CrO4) dissolves in acidic solution to generate strongly oxidizing CrO42- ions: K2CrO4(s) + H3O+(aq) â†' 2K+(aq) + HCrO4-(aq) + H2O(l) 2HCrO4-(aq) + CrO42-(aq) + H2O(l) â†' 3H+(aq) + 2Cr3+(aq) +

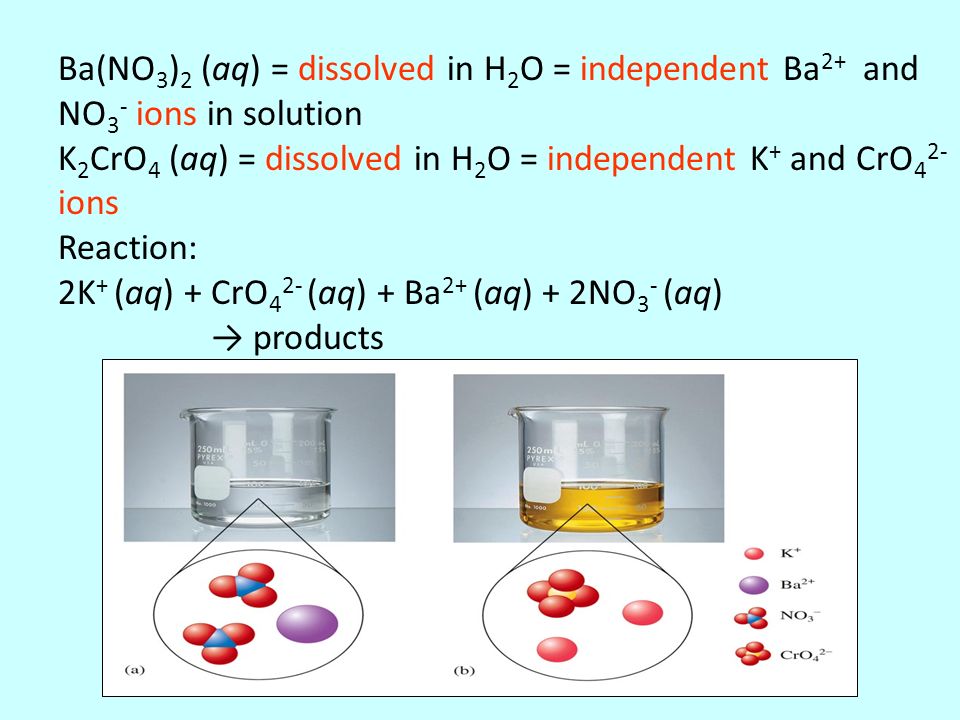
SOLVED: C.BaCl2 (aq) + K2CrO4 (aq) -> BaCrO4 + 2KCl (aq) 2CrO4^2- (aq) + 2H+ (aq) -> Cr2O7^2- (aq) + H2O Place approximately 5 mL of 0.1 M BaCl2 (barium chloride) solution
Q117) The set of numerical coefficients that balances the equation K2CrO4 + HCl → K2Cr2O7 + kCl + H2O is kerala CEE 2001 d) 2, 2, 1, 2, 1
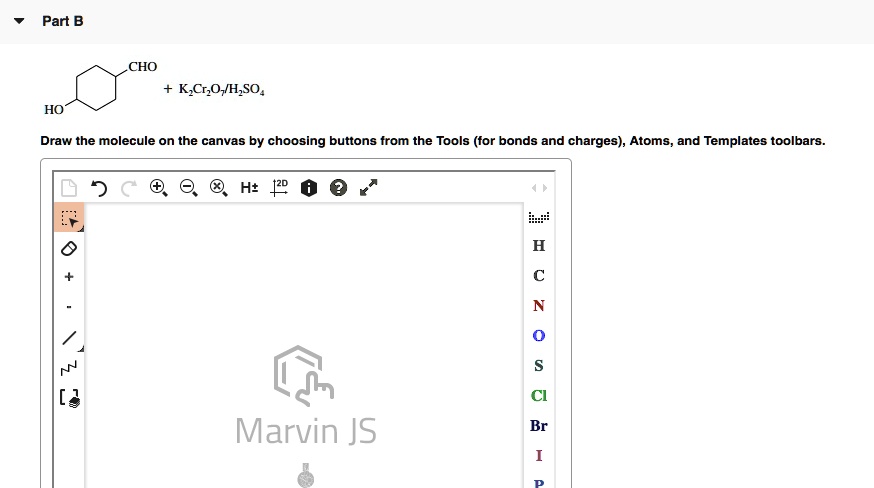
SOLVED: CHO K2CrO4 H2SO4 H2O Draw the molecule on the canvas by choosing buttons from the Tools (for bonds and charges), Atoms, and Templates toolbars. H2O E Marvin JS Br