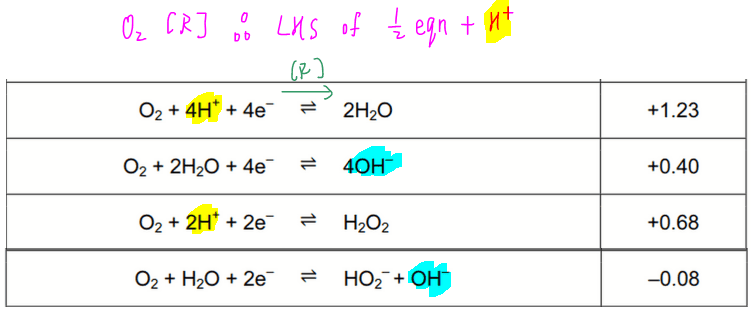CO2+H2=CO+H2O, 1 mole of CO2 and 2 moles of H2 are placed in a 2L container. At equilibrium, the concentration of CO is 0.28 mol/L. What is the equilibrium constant Kc for

Mechanistic studies of methanol synthesis over Cu from CO/CO2/H2/H2O mixtures: The source of C in methanol and the role of water - ScienceDirect

Consider the following reaction at certain temperature: H2O(g)+CO2(g) equilibrium to H2(g)+CO2(g) Some molecules of H2O and CO are placed in a 1.0 L container as shown below. When equilibrium is reached, how

Question⬇️ Does “moles of hydrogen gas” refer to moles of H2? Meaning that 2H2 is 2 moles of hydrogen gas? : r/chemhelp
![SOLVED: Determine the equilibrium-constant expression for the reaction: CuO(s) + H2(g) ⇌ Cu(l) + H2O(l) ? A. K = [H2O]/[CuO] B. K = [Cu][H2O]/[H2][CuO] C. K = 1/[H2] D. K = [H2]/[Cu] SOLVED: Determine the equilibrium-constant expression for the reaction: CuO(s) + H2(g) ⇌ Cu(l) + H2O(l) ? A. K = [H2O]/[CuO] B. K = [Cu][H2O]/[H2][CuO] C. K = 1/[H2] D. K = [H2]/[Cu]](https://cdn.numerade.com/ask_previews/69a6a069-64fe-4b52-b6aa-a926786b3f81_large.jpg)
SOLVED: Determine the equilibrium-constant expression for the reaction: CuO(s) + H2(g) ⇌ Cu(l) + H2O(l) ? A. K = [H2O]/[CuO] B. K = [Cu][H2O]/[H2][CuO] C. K = 1/[H2] D. K = [H2]/[Cu]
Consider the following reactions (i)H+(aq) + OH (aq) = H2O(l),△ H = X1kJ mol 1 (ii) H2( g) +1/202(g)= H20(l): △ H= X2 kJ mol 1 (iii) CO2(g)+ H2(g) = CO(g) +

Reaction of CO, H2O, H2 and CO2 on the clean as well as O, OH and H precovered Fe(100) and Fe(111) surfaces - Catalysis Science & Technology (RSC Publishing)

Illustration De La Formule Chimique Composée De Molécules Avec Le Concept D'éducation Icône Icône Chimie Et Sciences, Illustration Vectorielle, Diagramme. H2, H2O, NH2. Banque D'Images et Photos Libres De Droits. Image 73244621

Water formation on interstellar silicates: the role of Fe2+/H2 interactions in the O + H2 → H2O reaction | QuantumGrain

Direct production of H2O2 from H2 and O2 in a biphasic H2O/scCO2 system over a Pd/C catalyst: Optimization of reaction conditions - ScienceDirect

calculate the equilibrium constant of H2 + O2 gives us H2O + CEO at 13957 if the equilibrium constant 135 - Chemistry - Equilibrium - 13886927 | Meritnation.com
An equilibrium mixture, CO(g) + H2O(g) ⇋ CO2 (g) + H2 (g), present in a vessel of one litre capacity at 1000 K - Sarthaks eConnect | Largest Online Education Community

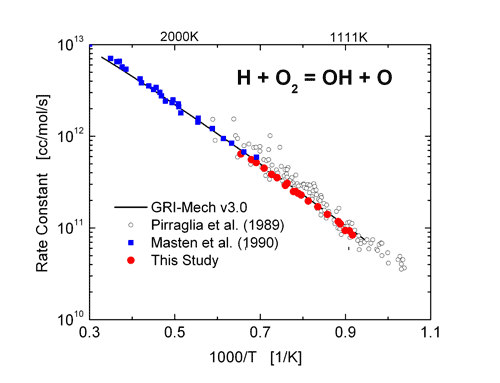





![PDF] Two triple points in the H2O–H2 system† | Semantic Scholar PDF] Two triple points in the H2O–H2 system† | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0e3a3f978107aae0e7b1ab47beca2a2ce22bd231/3-Figure2-1.png)


![PDF] Two triple points in the H2O–H2 system† | Semantic Scholar PDF] Two triple points in the H2O–H2 system† | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0e3a3f978107aae0e7b1ab47beca2a2ce22bd231/2-Figure1-1.png)