Balance KMnO4 + H2C2O4 + H2SO4 gives rise to K2SO4 + Mnso4 + CO2 + H2O using the alternate method of balancing

Acid base report - Title: acid-base titrations with lactic acid. Goal and method: determine the - Studeersnel

Balance the following chemical equationin the acidic medium by oxidationnumber method MnO4(aq) + C2H2O4(aq) - Brainly.in

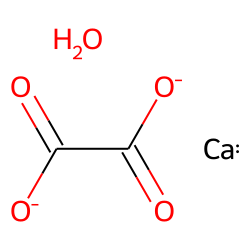
Oxalic acid (C2H2O4) is a diprotic acid. Write the two ionization reactions for C2H2O4 to C2O4^{2-}. | Homework.Study.com

Enthalpy of Formation of C2H2O4 (Oxalic Acid) from High-Level Calculations and the Active Thermochemical Tables Approach | The Journal of Physical Chemistry A