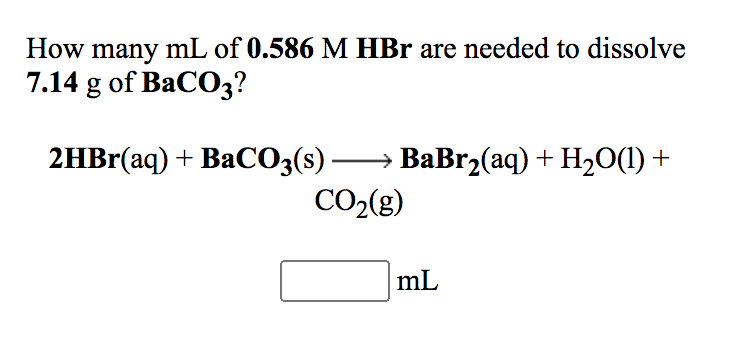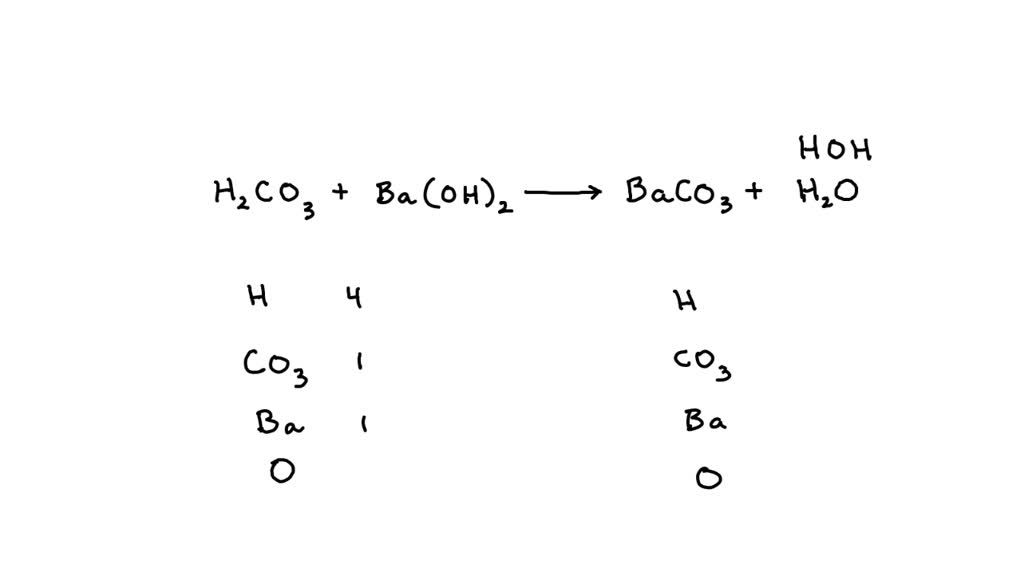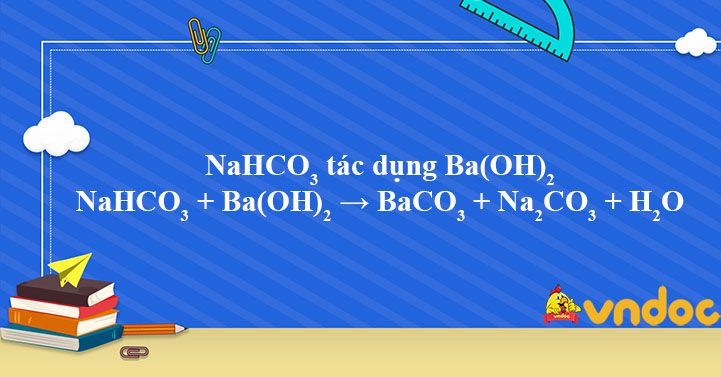SOLVED: (b) The Ksp of barium carbonate, BaCO3, is 2.58×10^(-9). Calculate the molar solubility, S, of this compound. S= (c) A 0.450 g sample of impure CaCO3(s) is dissolved in 50.0 mL

Synthetic pathway of 1. a) NH2OH·HCl, BaCO3, Pd/C, N2H4·H2O, reflux in... | Download Scientific Diagram

24.Write equilibrium constant expression the following reac (i) BaCO3 (8) ---------- BaO(s) + CO2 (g).
Calculate the mass of BaCO3 produced when excess CO2 is bubbled through a solution containing 0.205 moles of Ba(OH)2. - Sarthaks eConnect | Largest Online Education Community

Solve 53 sum 200 m' solution molarity of tho volumo ot CO, at S T P on heat'0 9 (2) - Chemistry - - 12898155 | Meritnation.com
Trajectory and timescale of oxygen and clumped isotope equilibration in the dissolved carbonate system under normal and enzymati
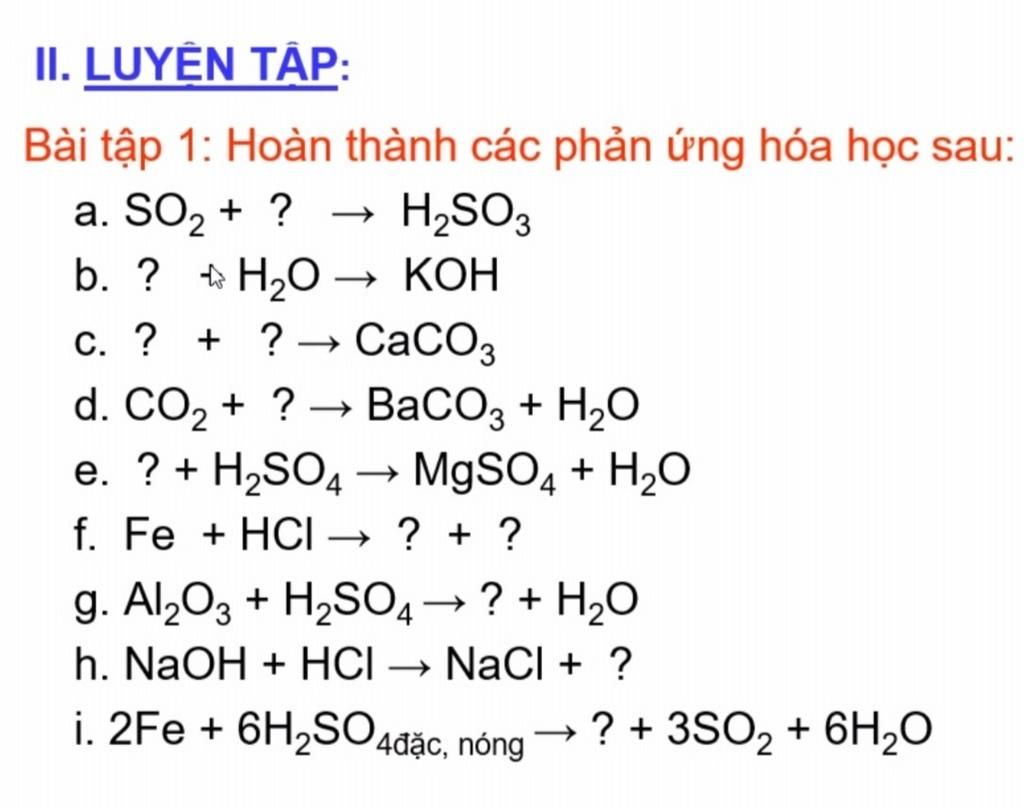
I1. LUYEΝ ΤΑΡ: Bài tập 1: Hoàn thành các phản ứng hóa học sau: a. SO, + ? → H2SO3 b. ? + H2O → KOH C. ? + ? СаСОз d. CO2 + ? → BaCO3 + H2O e. ? + H2SO4 f.
Solved: The chemical equation here describes a reaction between barium carbonate and nitric acid. [algebra]

a. SO2 + ? → H2SO3 b. ? +H2O → KOH c. ? + ?→ CaCO3 d. CO2 + ? → BaCO3 + H2O e. ? + H2S04 → MgSO4 + H2O f. Fe + HCl– → ? + ? g. Al203 + H2SO4 → ? + H2O h. N

Vidéo de question : Identifier la substance qui contient à la fois des liaisons ioniques et des liaisons covalentes | Nagwa

Question Video: Determining the Products of the Neutralization Reaction of Barium Hydroxide Ba(OH)₂ with Carbonic Acid H₂CO₃ | Nagwa

Date twe of bor WYSICAL SUENCE Works Balance the following equations: 1. AL + N2 - AIN 2. Fe + 02 - Fe3O4 Caco - CaO + CO2 NH.NO, N2O + H2O

SOLVED: Equation: Ba(OH)2(aq) CO2(g) BaCO3(s) H2O(l) Total ionic: Ba2+(aq) + 2OH-(aq) + CO2(g) â†' BaCO3(s) + H2O(l) Net ionic: Ba2+(aq) + CO2(g) â†' BaCO3(s) + H2O(l)

Cho sơ đồ các phản ứng theo đúng tỉ lệ mol:(a) X(t0) $ \to $ Y + CO2(b) Y + H2O $ \to $ Z(c) T + Z $ \to $ R + X + H2O(d?
Heritage | Free Full-Text | Characterization of Barium Hydroxide Used as Consolidating Agent for Monumental Surfaces in Venice

PPT - What is the difference between a chemical reaction and physical change? PowerPoint Presentation - ID:5813241






![ANSWERED] BaCO3 + 2HNO3 → Ba(NO3)2 + CO₂ + H₂O What ... - Organic Chemistry - Kunduz ANSWERED] BaCO3 + 2HNO3 → Ba(NO3)2 + CO₂ + H₂O What ... - Organic Chemistry - Kunduz](https://media.kunduz.com/media/answer/raw/20220422054128757632-4413054.jpg?type=wm)