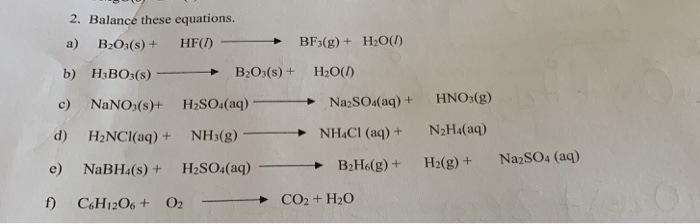
PDF) Phase equilibrium study in the CaO–K2O–B2O3–H2O system at 25°C | Laszlo Csetenyi - Academia.edu

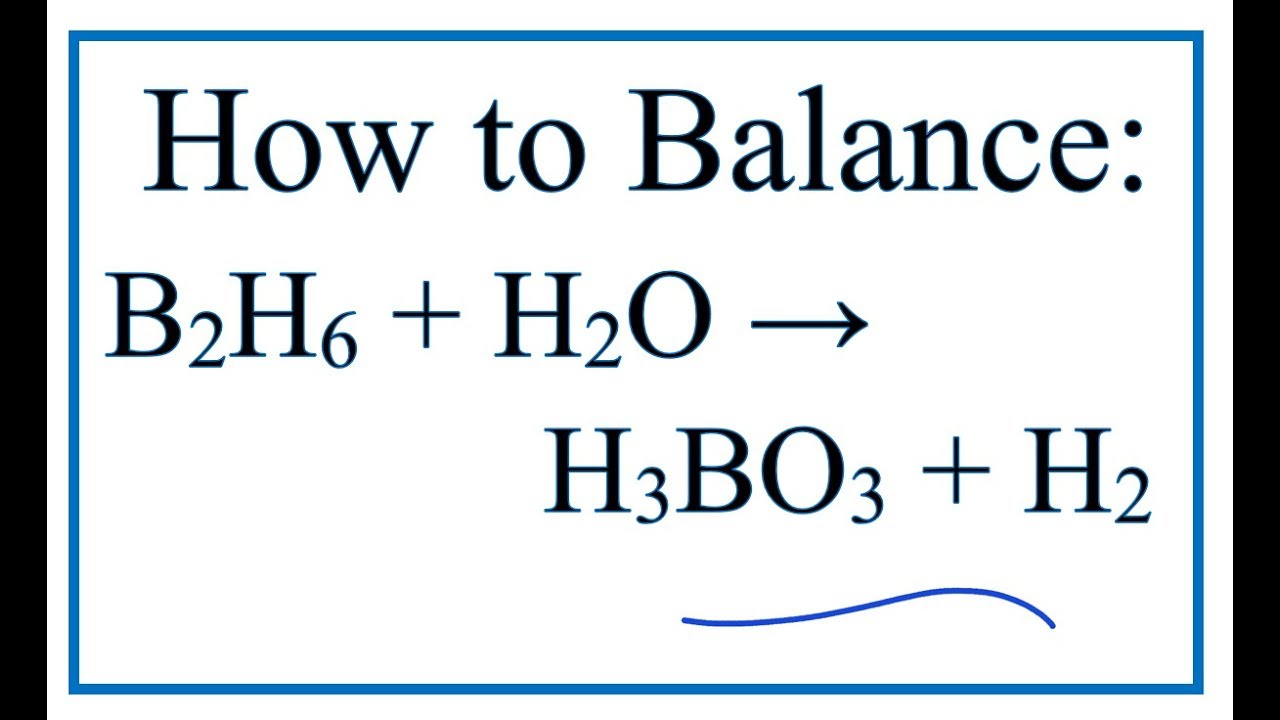
B2O3 + 3 H2O ---> 2 H3BO3 If diboron trioxide is reacted with water, the product is boric acid. What mass of boric acid is obtained from the full reaction. - ppt video online download
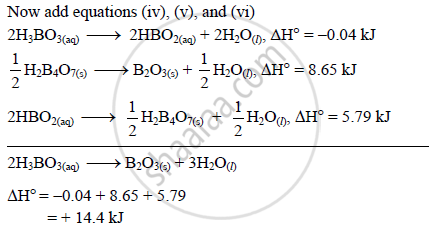
Calculate ∆H° for the Following Reaction: 2h3bo3(Aq) → B2o3(S) + 3h2o(L) Given That, - Chemistry | Shaalaa.com

Novel 4ZnO⋅B2O3⋅H2O:Ln3+/HTC (where Ln = Eu or Tb) phosphors: Synthesis, morphology and luminescence properties | Journal of Materials Research | Cambridge Core

Experimental and calculated phase diagrams in the system Na2O-B2O3-H2O... | Download Scientific Diagram

B2O3 + 3 H2O ---> 2 H3BO3 If diboron trioxide is reacted with water, the product is boric acid. What mass of boric acid is obtained from the full reaction. - ppt video online download

B2O3 + 3 H2O ---> 2 H3BO3 If diboron trioxide is reacted with water, the product is boric acid. What mass of boric acid is obtained from the full reaction. - ppt video online download
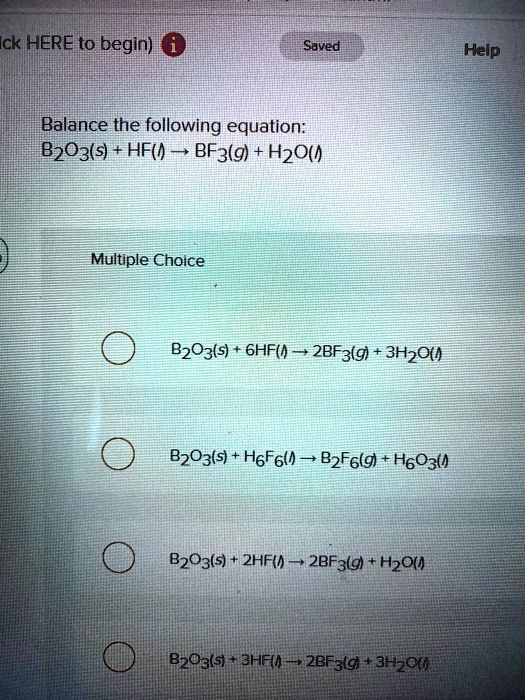
SOLVED: Click HERE to begin. Saved Help Balance the following equation: B2O3(s) + HF(g) â†' BF3(g) + H2O(l) Multiple Choice B2O3(s) + 6HF(g) â†' 2BF3(g) + 3H2O(l) B2O3(s) + 6HF(g) â†' B2F6(g) +